What's the difference between sugar, other natural sweeteners and artificial sweeteners? A food chemist explains sweet science
The Conversation
By Kristine Nolin, University of Richmond
A quick walk down the drink aisle of any corner store reveals the incredible ingenuity of food scientists in search of sweet flavors. In some drinks you’ll find sugar. A diet soda might have an artificial or natural low-calorie sweetener. And found in nearly everything else is high fructose corn syrup, the king of U.S. sweetness.
I am a chemist who studies compounds found in nature, and I am also a lover of food. With confusing food labels claiming foods and beverages to be diet, zero-sugar or with “no artificial sweeteners,” it can be confusing to know exactly what you are consuming.
So what are these sweet molecules? How can cane sugar and artificial sweeteners produce such similar flavors? First, it is helpful to understand how taste buds work.

Taste buds and chemistry
The “taste map” – the idea that you taste different flavors on different parts of your tongue – is far from the truth. People are able to taste all flavors anywhere there are taste buds. So what’s a taste bud?
Taste buds are areas on your tongue that contain dozens of taste receptor cells. These cells can detect the five flavors – sweet, sour, salty, bitter and umami. When you eat, food molecules are dissolved in saliva and then washed across the taste buds, where they bind to the different taste receptor cells. Only molecules with certain shapes can bind to certain receptors, and this produces the perception of different flavors.
Molecules that taste sweet bind to specific proteins on the taste receptor cells called G-proteins. When a molecule binds these G-proteins, it triggers a series of signals that are sent to the brain where it is interpreted as sweet.
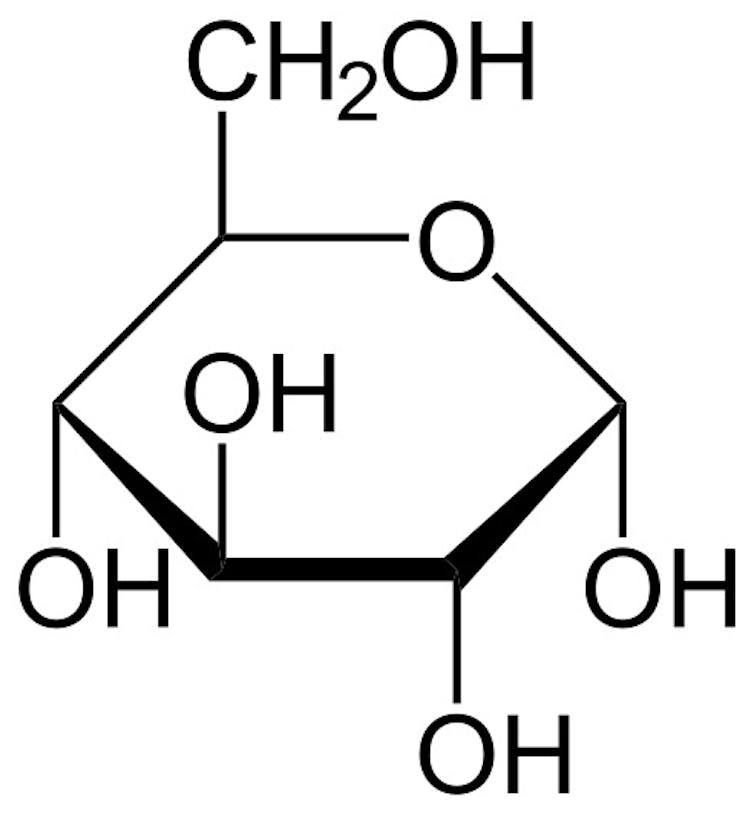
Natural sugars
Natural sugars are types of carbohydrates known as saccharides that are made of carbon, oxygen and hydrogen. You can imagine sugars as rings of carbon atoms with pairs of oxygen and hydrogen attached to the outside of the rings. The oxygen and hydrogen groups are what make sugar sticky to the touch. They behave like Velcro, sticking to the oxygen and hydrogen pairs on other sugar molecules.
The simplest sugars are single-molecule sugars called monosaccharides. You’ve probably heard of some of these. Glucose is the most basic sugar and is mostly made by plants. Fructose is a sugar from fruit. Galactose is a sugar in milk.
Table sugar – or sucrose, which comes from sugar cane – is an example of a dissacharide, a compound made of two monosaccharides. Sucrose is formed when a glucose molecule and a fructose molecule join together. Other common dissacharides are lactose from milk and maltose, which comes grains.
When these sugars are eaten, the body processes each of them slightly differently. But eventually they are broken down into molecules that your body converts into energy. The amount of energy from sugar – and all food – is measured in calories.

High fructose corn syrup
High fructose corn syrup is a staple of U.S. foods, and this hybrid sugar sweetener needs a category all on its own. High fructose corn syrup is made from corn starch – the main carbohydrate found in corn. Corn starch is made of thousands of glucose molecules bonded together. At an industrial scale, the starch is broken into individual glucose molecules using enzymes. This glucose is then treated with a second enzyme to convert some of it into fructose. Generally, high fructose corn syrup is roughly 42%-55% fructose.
This blend is sweet and cheap to produce but has a high calorie content. As with other natural sugars, too much high fructose corn syrup is bad for your health. And since most processed foods and drinks are packed full of the stuff, it is easy to consume too much.

Natural nonsugar sweeteners
The second category of sweeteners could be defined as natural nonsugar sweeteners. These are food additives such as stevia and monk fruit, as well as natural sugar alcohols. These molecules aren’t sugars, but they can still bind to the sweet receptors and therefore taste sweet.
Stevia is a molecule that comes from the leaves of the Stevia redaudiana plant. It contains “sweet” molecules that are much larger than most sugars and have three glucose molecules attached to them. These molecules are 30 to 150 times sweeter than glucose itself. The sweet molecules from monk fruit are similar to stevia and 250 times sweeter than glucose.
The human body has a really hard time breaking down both stevia and monk fruit. So even though they’re both really sweet, you don’t get any calories from eating them.
Sugar alcohols, like sorbital, for example, are not as sweet as sucrose. They can be found in a variety of foods, including pineapples, mushrooms, carrots and seaweed, and are often added to diet drinks, sugar-free chewing gum and many other foods and drinks. Sugar alcohols are made of chains of carbon atoms instead of circles like normal sugars. While they are composed of the same atoms as the sugars, sugar alcohols are not absorbed well by the body so they are considered low-calorie sweeteners.

Artificial sweeteners
The third way to make something sweet is to add artificial sweeteners. These chemicals are produced in labs and factories and are not found in nature. Like all things that taste sweet, they do so because they can bind to certain receptors in taste buds.
[Over 140,000 readers rely on The Conversation’s newsletters to understand the world. Sign up today.]
So far, the U.S. Food and Drug Administration has approved six artificial sweeteners. The most well known are probably saccharin, aspartame and sucralose – better known as Splenda. Artificial sweeteners all have different chemical formulas. Some resemble natural sugars while others are radically different. They are usually many times sweeter than sugar – saccharin is an incredible 200 to 700 times sweeter than table sugar – and some of them are hard for the body to break down.
While a sweet dessert may be a simple pleasure for many, the chemistry of how your taste buds perceive sweetness is not so simple. Only molecules with the perfect combination of atoms taste sweet, but bodies deal with each of these molecules differently when it comes to calories.![]()
Kristine Nolin, Associate Professor of Chemistry, University of Richmond
This article is republished from The Conversation under a Creative Commons license. Read the original article.

